IVD MEDICAL DEVICES
Specialising in BD in-vitro diagnostic systems for cervical cancer screening and women's health — including liquid-based cytology, HPV genotyping and vaginal infection detection. READ MORE >CROSS-BORDER PROCUREMENT
Leveraging Hong Kong's international trade hub advantages to provide efficient cross-border sourcing, import/export compliance and end-to-end logistics for medical device clients worldwide. READ MORE >REGULATORY & COMPLIANCE
Expert guidance on NMPA registration, CE marking and import documentation — ensuring every product meets the full regulatory requirements of the target market. READ MORE >TECHNICAL SUPPORT
Comprehensive after-sales service covering on-site installation, operator training, application consultancy and ongoing technical support to maximise product performance. READ MORE >YEARS EXPERIENCE
GROUP COMPANIES
BRAND SUPPLIERS
REGIONAL OFFICES
Hong Kong Origins Investment Limited is a professional cross-border trade and industrial investment enterprise rooted in Hong Kong with a global reach, focusing on international trade and industry services for medical devices and laboratory products. Leveraging Hong Kong's position as an international financial and trade hub, we have built an efficient cross-border supply chain and value service platform.
With deep expertise in international trade, we provide clients worldwide with comprehensive import/export solutions through professional supply chain management and compliance services — covering product selection, cross-border procurement, international logistics, customs clearance and local delivery. Backed by a mature global cooperation network, we have established long-term strategic partnerships with leading brands and quality manufacturers, continuously delivering high-quality, well-adapted products and services to our clients.
With over a decade of professional experience in international medical device trade, we deliver comprehensive, compliant and efficient solutions that connect world-class manufacturers with healthcare and research institutions globally.

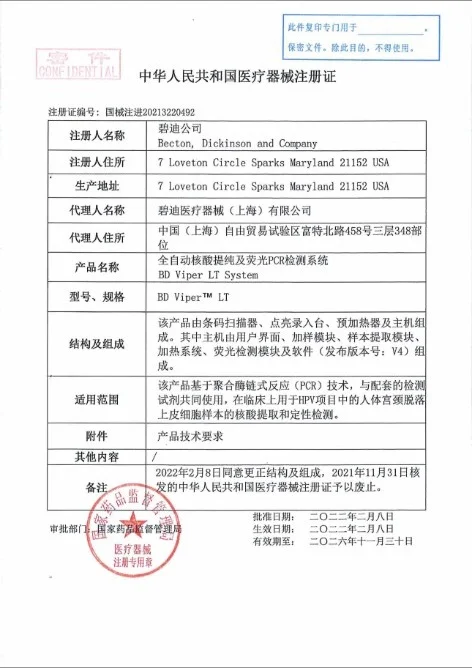
NMPA-issued registration certificate for medical devices distributed in the Chinese market.

In-vitro diagnostic reagent registration approved by the National Medical Products Administration.

Class I medical device filing certificate issued by the NMPA, confirming regulatory compliance.
We start with an in-depth conversation to understand your procurement goals, budget constraints and delivery timeline.
Our specialists craft a tailored product plan with detailed quotation, lead time and supplier recommendation.
We engage verified global suppliers and execute cross-border purchasing on your behalf at competitive terms.
Every batch undergoes rigorous inspection against international standards before leaving the supplier warehouse.
We manage full-chain shipping, customs clearance and all import/export compliance documentation end to end.
Final-mile delivery, on-site installation and commissioning, with dedicated ongoing after-sales support.

Full BD SurePath™ LBC system deployment — covering product selection, NMPA clearance, customs import, on-site installation and clinical staff training.

End-to-end procurement of BD Viper LT™ + Onclarity™ HPV system, providing co-testing with existing LBC workflow and regulatory compliance documentation.

Coordinated multi-site deployment of BD Affirm™ VPIII vaginal infection detection systems across an outpatient network, including staff training and reagent supply chain setup.